Article Text
Abstract
Background Several lines of evidence suggest that atrial fibrillation (AF) may be a consequence of vascular disease. We investigated the relationship between cardio-ankle vascular index (CAVI), a new index of arterial stiffness, and the presence of paroxysmal AF (PAF).
Methods and results 181 outpatients (91 patients with PAF and 90 age- and gender-matched subjects without PAF) were analysed for their sinus rhythm. The CAVI was significantly higher in patients with PAF than in subjects without PAF (9.0±1.0 vs 8.7±0.8, p<0.01). In all subjects, the CAVI was significantly correlated with the left ventricular mass index (r=0.30, p<0.01), left atrial diameter (r=0.22, p<0.01), and augmentation index, a parameter of wave reflection (r=0.32, p<0.01), in addition to age, systolic blood pressure and pulse pressure. Logistic analysis demonstrated that the CAVI was independently associated with PAF even after adjustment for confounding factors. The adjusted OR of PAF was 1.8 for each unit increase in the CAVI (p=0.01).
Conclusions Our finding suggests that increased arterial stiffness may be involved in the maintenance of AF.
- Peripheral Vascular Disease
- Hypertension
Statistics from Altmetric.com
Background
Atrial fibrillation (AF) is the most common chronic arrhythmia in adults and is associated with an increased risk of mortality from cardiovascular events.1 ,2 Although several clinical risk factors for AF have been identified,3––6 further evaluation of modifiable risk factors is required to prevent the development of AF. A cohort study showed that carotid intima-media thickness is associated with the new onset of AF.7 In a case-control study, carotid intima-media thickness as well as arterial stiffness assessed by carotid-femoral pulse wave velocity (PWV) was associated with lone AF.8 Increased arterial stiffness, which leads to increased cardiac pressure load, ventricular hypertrophy and left atrial enlargement, could be the perpetuation of AF.9––11 Thus, the interaction of vascular function and cardiac load may play a critical role in the maintenance of AF.
The cardio-ankle vascular index (CAVI) has been developed for the quantitative evaluation of vascular wall stiffness in the aorta, femoral arteries and tibial artery by measuring PWV and blood pressure.12 Indeed, the CAVI correlates with other cardiovascular risk markers, such as intima-media thickening and coronary atherosclerosis, thus reflecting the degree of atherosclerotic change.13 Arterial stiffness affects wave reflection, which results when a forward pressure wave created by ventricular contraction encounters a site of impedance.14 ,15 Thus, increased arterial stiffness can induce left ventricular (LV) hypertrophy and left atrial dilation.16
We previously showed that the augmentation index is significantly associated with the presence of paroxysmal AF.17 From this, we hypothesised that the CAVI may also be associated with AF. As an examination of the CAVI in patients with chronic AF is technically problematic, we analysed the CAVI in subjects with and without paroxysmal AF in this study.
Methods
Ethics statement
The experimental procedures and potential risks were explained prior to the study. The Institutional Review Board of Okayama University and Sumitomo Besshi Hospital approved this study and informed consent was obtained from each subject. The study conformed to the principles of the Declaration of Helsinki.
Subjects
We enrolled 91 outpatients with a history of paroxysmal AF and 90 age- and gender-matched subjects without a history of paroxysmal AF, all of whom visited Sumitomo Besshi Hospital or Okayama University Hospital from October 2009 to May 2011. Paroxysmal AF was diagnosed according to the American College of Cardiology/American Heart Association/European Society of Cardiology OR ACC/AHA/ESC 2006 Guidelines.18 Paroxysmal AF was documented at least once with 24 h holter monitoring or at clinic 3 months before the enrolment. All subjects were in sinus rhythm at the time of measuring the CAVI and augmentation index. Patients were excluded from the study if they had any of the following conditions: (1) long standing persistent AF; (2) renal insufficiency (serum creatine >2.0 mg/mL); (3) acute coronary syndrome; (4) severe valvular disease and prosthetic valves; (5) decreased LV systolic function (LV ejection fraction <50%); or (6) ankle-brachial index <0.9.
CAVI measurement
The CAVI was calculated automatically using a VaSera VS-1000 Vascular Screening System (Fukuda Denshi, Tokyo, Japan) from the blood pressure and PWV, while simultaneously monitoring the electrocardiogram and heart sounds.12 ,19 PWV was calculated by dividing the distance from the aortic valve to the ankle artery by the sum of the time between the aortic valve closing sound and the notch of the brachial pulse wave and the time between the rise of the brachial pulse wave and the ankle pulse wave. The CAVI was determined by the following equation: CAVI=a[(2ρ/ΔP) × ln(Ps/Pd)×PWV2]+b, where Ps and Pd are the systolic and diastolic blood pressure, respectively; PWV is between the heart and ankle; ΔP is Ps − Pd; ρ is blood density; and a and b are constants. The average of the right and left CAVI values was used for analysis.
Blood pressure measurements and pulse wave analysis
After a 5-min rest and with the subject seated, brachial blood pressure was measured using an automatic cuff oscillometric device and the average of two readings was used. Next, the radial pulse wave was examined at the wrist using applanation tonometry with a high-fidelity micromanometer (HEM-9000AI, Omron Healthcare Co., Kyoto, Japan), as previously described.16
Transthoracic echocardiography
All echocardiographic data were obtained by experienced sonographers and interpreted by an experienced staff echocardiographer. Comprehensive examinations were performed including M-mode, 2D, conventional Doppler and colour Doppler echocardiography. Measurements were made according to the guidelines of the American Society of Echocardiography.20 The left atrial dimensions were assessed by M-mode echocardiography and LV mass was calculated using the Penn convention.21 The LV mass index was calculated by dividing the LV mass by the body surface area. The LV ejection fraction was also examined as a measure of cardiac function.
Statistical analysis
Data are summarised as mean±SD for quantitative variables. Categorical variables were compared between groups by χ2 analysis, and continuous variables were compared between two groups by unpaired t test. Bivariate correlation was assessed using Pearson's correlation analysis. Multiple logistic regression analysis was performed to evaluate the relationships between paroxysmal AF and increasing CAVI, aspects of blood pressure and echocardiographic parameters. The sample size was determined on the basis of the estimated CAVI values reported in our other study.16 We assumed a CAVI of 8.8±1.0 (mean±SD) in the control group and 9.0±1.0. Using a two-sided test for differences in independent binominal proportions with an α level of 0.05, we calculated that 172 patients (86 in each group) would have 90% power to detect a difference in the CAVI between two groups. Differences at p<0.05 were considered significant. Data were analysed using SPSS V.17.0 for Windows (SPSS Inc., Chicago, Illinois, USA).
Results
Table 1 shows the baseline characteristics of the AF and control groups. There were no differences between the groups with regard to age, gender, height, body mass index, prevalence of hypertension and diabetes, whereas the control group included a lower percentage of subjects with hyperlipidaemia than the paroxysmal AF group. Systolic blood pressure, diastolic blood pressure, mean arterial pressure and pulse pressure were comparable between the two groups (table 2). The heart rate in patients with paroxysmal AF was lower than that in patients without paroxysmal AF (table 2). Regarding the drugs administered, the use of β-blockers, digitalis and antiarrhythmic drugs was higher in patients with paroxysmal AF than in those without paroxysmal AF. There were no significant differences between the two groups in the use of ACE inhibitors and/or angiotensin receptor blockers, calcium channel blockers, and statins. As for echocardiographic parameters, the left atrial diameter was significantly larger in patients with paroxysmal AF.
Patient characteristics
Haemodynamic and echocardiographic parameters
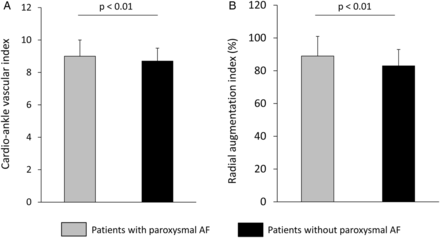
The CAVI value in patients with paroxysmal AF was significantly higher than that in subjects without paroxysmal AF (9.0±1.0 vs 8.7±0.8, p<0.01; figure 1). The radial augmentation index value for patients with paroxysmal AF was significantly higher than for subjects without paroxysmal AF (89±12% vs 83±10%, p<0.01; figure 1).
Cardio-ankle vascular index (A) and radial augmentation index (B) values in patients with or without paroxysmal atrial fibrillation (AF). Data are presented as mean+SD.
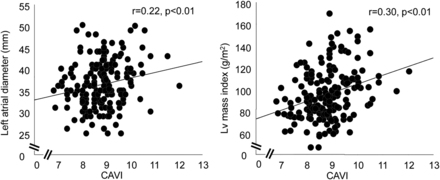
Table 3 shows the associations between the CAVI and other parameters of blood pressure, biochemical markers and echocardiography. The CAVI was significantly and positively correlated with age, systolic blood pressure and pulse pressure, and was significantly and negatively correlated with high-density lipoprotein cholesterol. Regarding echocardiographic parameters, the CAVI was significantly and positively correlated with LV mass index and left atrial diameter (figure 2). Furthermore, the CAVI was significantly associated with radial augmentation index (figure 3).
Association between the CAVI and other parameters
Correlation of cardio-ankle vascular index with left ventricular mass index and left atrial diameter.
Association between cardio-ankle vascular index and radial augmentation index in all subjects.
We next carried out a multivariate analysis to evaluate the relative associations of the CAVI, individual blood pressure parameters, LV mass index and left atrial diameter with paroxysmal AF in a model adjusted for age, gender, heart rate, and use of antihypertensive and antiarrhythmic drugs. In this model, increases in the CAVI and left atrial diameter significantly associated with an increased risk of paroxysmal AF (table 4). Multiple logistic analysis revealed that the OR of paroxysmal AF was elevated ∼1.8 for each unit increase in the CAVI (table 4).
Association of paroxysmal atrial fibrillation with the CAVI, blood pressure parameters and echocardiographic parameters
Discussion
This study showed that an increase in arterial stiffness, as measured by the CAVI, was independently and positively associated with paroxysmal AF. Even after adjusting for other relevant factors, patients with paroxysmal AF had significantly higher CAVI values than did those without paroxysmal AF. These findings suggest a potential role of arterial structural and functional remodelling in the pathogenesis of AF.
Our data are in good agreement with those from a previous study showing increased arterial stiffness assessed by carotid-femoral PWV in patients with lone AF.8 Ours is the first study to evaluate the association between the CAVI and AF. Regarding the mechanism by which an increase in arterial stiffness may increase paroxysmal AF, one possibility is that enhanced LV load and hypertrophy caused by an increase in the augmentation index may elevate pressure and enlarge the left atrium, thus maintaining AF. This study showed a positive correlation between the CAVI and the radial augmentation index, suggesting that an increase in arterial stiffness affects LV afterload. In line with our previous studies,16 ,22 this study also found that the CAVI was positively associated with the LV mass index and left atrial size. Another potential mechanism for the association between arterial stiffness and AF is that increased arterial stiffness predisposes individuals to a generalised cardiovascular inflammatory response.23 ,24 In fact, a case-control study showed that C reactive protein is elevated in patients with AF,25 and high sensitivity C reactive protein had significant positive association between carotid-femoral PWV even in healthy population.26
The CAVI is a widely used non-invasive measure of arterial stiffness with the benefit of being independent of blood pressure.12 The CAVI tends to be higher in patients with hypertension than in non-hypertensive patients13 and this study also showed a correlation between the CAVI and systolic blood pressure; this, however, does not necessarily mean that the CAVI is dependent on blood pressure. In fact, Shirai et al27 reported that the β1-adrenoceptor blocker metoprolol lowers systolic blood pressure but does not decrease the CAVI. Considering the interaction between arterial stiffness and paroxysmal AF, we need to identify the arterial stiffness which is not interfered by blood pressure at the time of measuring. The independence of the CAVI from occasional blood pressure may be advantageous compared with carotid-femoral PWV. Hypertension treatment is one of the most important factors in the prevention of AF.28 Therefore, using the CAVI to evaluate the effectiveness of hypertensive treatment on vascular properties could further help in prevention of AF. Additional larger-scale prospective studies are warranted to determine the applicability of the CAVI to assessing risk of AF in the general population.
The present study had several limitations. First, data on smoking, which is known to affect the CAVI, were not obtained from the participants. We cannot rule out the possibility that there were different proportions of smokers in the AF and control groups. Second, it is known that the use of antihypertensive influences the CAVI. We analysed the relationship between the CAVI and paroxysmal AF in subjects receiving prescription medications; therefore, we cannot reject a possible influence of the medications on paroxysmal AF. Third, because our study was cross-sectional, we could not clearly establish a causal relationship between the CAVI and paroxysmal AF. In addition, participants were enrolled from only two sites. Some selection bias cannot be denied. Prospective multicentre studies are necessary to resolve this issue.
Conclusions
In conclusion, this study demonstrated that an increase in the CAVI is independently associated with paroxysmal AF. Our findings indicated that the CAVI might represent a modifiable risk marker for AF.
References
Footnotes
-
Contributors TM, MD, YN, YO, KS, SK and YN conceived the study, and participated in its design and coordination and helped to draft the manuscript. HI was involved in drafting the manuscript or revising it critically.
-
Competing interests None.
-
Patient consent Obtained.
-
Ethics approval The Institutional Review Board of Okayama University.
-
Provenance and peer review Not commissioned; externally peer reviewed.